Background: PD-L1 expression has been explored in solid tumours and in classical Hodgkin's lymphoma, however its expression in acute leukemias has been minimally studied. PD-L1 over-expression in AML blasts has previously been associated with worse outcomes in patients with mutated FTL3 genes, and positively correlated with TP53 expression. Over-expression of PD-L1 may contribute to failed response to hypo-methylating agents, leading to the rationale behind combination therapy with immune-checkpoint inhibitors. Despite evidence that the PD-1/PDL-1 axis may promote myeloid neoplasms by modulating immune escape, clinical trials studying the role of immunotherapy in MDS/AML have produced disappointing results. The lack of evidence to support the role of PD-1-targeted therapy in AML may be due to lack of identification of the subset of patients who may benefit from immunotherapy. We previously demonstrated that PD-L1 expression is significantly enhanced in patients with JAK2/STAT mutations compared to patients with wild-type (WT) JAK2/STAT. Here, we investigate the clinical correlate of our findings and compare clinical outcomes of AML patients with respect to PD-L1 expression.
Methods: Immunohistochemical staining for PD-L1 was previously performed on bone marrow biopsies of 31 AML patients, including 11 patients with JAK2/STAT mutations (nine with JAK2 V617F exon 14 mutations, one with JAK2 I540-E543delinsRG exon 12 mutation and one with STAT5B N642H mutation) and 20 patients with WT JAK2/STAT. PD-L1 CPS was calculated by summation of all PD-L1-stained cells divided by total number of tumour cells, and multiplied by 100. Retrospective chart review was performed to extract data regarding patient demographics, cytogenetics, mutations, treatment course, as well as clinical outcomes. Low and high PD-L1 expression was defined as CPS score <10 and > 20 respectively. Kaplan-Meier analysis was used for analysing overall survival (OS) and event-free survival (EFS), with statistical significance determined using a 2-tailed log-rank test. EFS was defined as duration of time between diagnosis and the first occurrence of either going off protocol induction without achieving complete remission (CR), relapse from CR, or death due to any cause. Patients who were alive at the time of study analysis were censored at date of last follow-up.
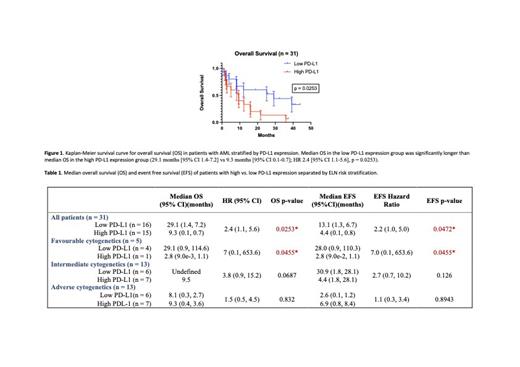
Results: A total 15 patients demonstrated high PD-L1 expression (lowest PD-L1 CPS 20), including four patients with WT JAK2/STAT and all 11 patients with mutant JAK2/STAT. 16 patients demonstrated low PD-L1 expression (highest PD-L1 CPS 6). Only three patients in the low PD-L1 expression group never achieved complete remission compared to eight patients in the high PD-L1 expression group. Median OS in the low PD-L1 expression group was significantly longer than median OS in the high PD-L1 expression group (29.1 months [95% CI 1.4-7.2] vs 9.3 months [95% CI 0.1-0.7]; HR 2.4 [95% CI 1.1-5.6], p = 0.0253, Figure 1). Similarity, EFS was significantly longer in the low PD-L1 expression group (13.1 months [95% CI 1.3-6.7] vs. 4.4 months [95% CI 0.1-0.8]; HR 2.4 [95% CI 1.1-5.6], p = 0.0253). When analysis was separated by European LeukemiaNet (ELN) risk stratification, significant differences in OS and EFS were re-demonstrated in patients with favourable ELN risk, but not in patients with intermediate or adverse risks. When wild-type JAK2/STAT patients were analyzed separately, there was no significant differences between OS and EFS with respect to PD-L1 expression.
Conclusions: We previously demonstrated that PD-L1 is significantly up-regulated in AML patients who carry mutant JAK2/STAT when compared to WT JAK2. Using a proposed PD-L1 CPS cut-off score of 20, we demonstrated that high PD-L1 expression was associated with significantly worse outcomes with respect to OS and EFS. These results may indicate a therapeutic role of immunotherapy in the treatment of AML, particularly in patients with JAK2/STAT mutations.
Disclosures
Konopleva:Abbvie, Allogene Therapeutics, Cellectis, Forty Seven, Gilead Sciences, Genentech, Sanofi, MEI Pharma, Rafael Pharmaceuticals, Daiichi Sankyo Pharmaceutical, AstraZeneca Co., Menarini, Precision BioSciences.: Research Funding; Reata Pharmaceuticals.: Current holder of stock options in a privately-held company, Patents & Royalties; AbbVie, Forty Seven, Precision Biosciences, Gilead Sciences, Genentech, Janssen, Sanofi, MEI Pharma, Daiichi Sankyo Pharmaceutical, AstraZeneca Co., Menarini.: Consultancy. Mantzaris:Kite, a Gilead company: Honoraria. Shastri:Kymera Therapeutics: Honoraria, Research Funding; Gilead Sciences: Honoraria; Rigel Pharmaceuticals: Honoraria; Janssen Pharmaceuticals, Inc.: Consultancy, Honoraria. Gritsman:iOnctura: Research Funding; ADC Therapeutics: Research Funding. Verma:Prelude: Research Funding; Throws Exception: Current equity holder in private company; BMS: Research Funding; GSK: Research Funding; Incyte: Research Funding; Medpacto: Research Funding; Curis: Research Funding; Eli Lilly: Research Funding; Stelexis: Current equity holder in private company, Honoraria, Other: Scientific Advisor; Bakx: Current equity holder in private company, Other: Scientific Advisor; Novartis: Other: Scientific Advisor; Acceleron: Other: Scientific Advisor; Celgene: Other: Scientific Advisor; Janssen: Honoraria.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal